p-Anisaldehyde
-
- Category :
Intermediates/Pharmaceutical intermediates
- CAS NO : 123-11-5
- EC NO : 204-602-6
- Molecular Formula : C8H8O2
- Main Specifications : 99% min
- Synonyms : AnisaldehydeMetoxybenzaldehyd;aubepine;Anisaldehyde;4-Methoxybenzaldehyde;4-Methoxybenzylaldehyde;Anisic Aldehyde;p-methoxybenzaldehyde;4-methoxy-benzaldehyd;Anisal;Benzaldehyde,4-methoxy-;Methyl-p-oxybenzaldehyde;Obepin;p-Formylanisole;p-Methoxybenzafdehyde;PARA Anisaldehyde;PARA Anisic Aldehyde;P-Anisic Aldehyde;FEMA 2670;AUBE'PINE;Anisaldehyde,4-;Anisic Aldehyde-P;LABOTEST-BB LT00920037;4-Anisaldehyde;Benzaldehyde, methoxy-;
Package: 25kg,50kg 200kg
Uses : involving spices, pharmaceuticals, and industrial fields
Molecular Structure:
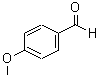
Product description:
What is the chemical of p-Anisaldehyde ?
Appearance: Colorless to pale yellow liquid, solidifies when cold, with a strong floral aroma similar to hawthorn ;
Assay:99%min by GC ;
IR Identity: conform to standard ;
HNMR: conform to standard ;
carbon spectrum: conform to standard ;
Water by K. F.:0.5% max or as per the customer’s request ;
Loss on drying:0.5% max. or as per the customer’s request ;
boiling point: 248 °C(lit.);
============================================================================
It has a wide range of applications, mainly involving spices, pharmaceuticals, and industrial fields:
Spice industry: as the main spice of hawthorn flower, sunflower flower, lilac and other flower scented essence, it is also used for modifying agents such as lily of the valley and osmanthus, as well as mixing vanilla, cream, chocolate and other food essence; In daily essence, it can be used as soap essence.
Pharmaceutical industry: as a synthetic intermediate for antimicrobial drugs (such as amoxicillin) and antihistamines.
Industrial application: Used as a brightener in cyanide free galvanizing process to improve anodic polarization and obtain a bright coating; Simultaneously used as an organic synthesis intermediate for the preparation of other chemicals
===========================================================================
The chemical synthesis routes mainly include the following methods:
Benzal-dehyde methylation method: Using benzal-dehyde as the raw material, it first reacts with a methylation reagent (such as dimethyl sulfate) to produce anisole, and then introduces chloromethyl through chloromethylation reaction. Finally, it is converted into aldehyde group through hydrolysis or salt formation of Urotropin. This route has a high yield and is suitable for large-scale production.
Phenol methylation method: Phenol is methylated to produce anisole, which is then subjected to the Sommelier reaction (chloromethylation hydrolysis) to obtain the target product. This route provides readily available raw materials, but attention should be paid to controlling side reactions.
P-Toluenol oxidation method: p-Toluenol is first methylated to p-methylphenyl ether, and then oxidized with oxidants (such as potassium dichromate, potassium permanganate, or manganese dioxide) to produce p-methoxybenzal-dehyde. This method has simple steps, but the choice of oxidant needs to balance cost and environmental impact.
