beta-Naphthyl ethyl ether
-
- Category :
Organic chemicals and Derivatives/Aroma compounds
- CAS NO : 93-18-5
- EC NO : 202-226-7
- Molecular Formula : C12H12O
- Main Specifications : 99% min
- Synonyms : Beta-naphthyl ethyl ether;Ethyl 2-naphthyl ether;Ethyl beta-naphthyl ether;Naphthalene, 2-ethoxy-;Nerolin bromelia;2-ethoxy-naphthalen;2-Ethoxynaphthalin;beta-Ethoxynaphthalene;b-Naphthol-athylather;Bromelia (compound);Ethyl beta-naphtholate;ethylbeta-naphtholate;Ethyl-naphthylether;Neroline bromelia;2-Naphthol ethyl ether;
Package: 25kg 50kg drum or bag
Uses : used as the flavoring ingredient of soap essence
Molecular Structure:
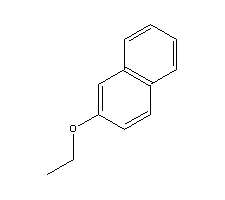
Product description:
What is the chemical of beta-Naphthyl ethyl ether?
Appearance: White crystals or white crystal flakes with orange blossom fragrance;
Assay: 99% min by HPLC ;
IR Identity: conform to standard ;
HNMR: conform to standard ;
carbon spectrum: conform to standard ;
Water by K. F.:0.5% max or as per the customer’s request ;
Loss on drying:0.5% max. or as per the customer’s request ;
Melting point: 282-284 ° C (atmospheric pressure, 760 mmHg);
===============================================================================
main purpose
It is used as the flavoring ingredient of soap essence (such as jasmine, orange blossom and cologne)
As a sweetener and floral fragrance, it is often used in conjunction with methyl naphthyl ketone, methyl ortho aminobenzoate, orange leaf oil, etc
It can be used to mix strawberry, berry, cherry, coffee, pomegranate, plum, black tea and other edible essence
In the field of medicine, it is the raw material for sodium oxynaproxen
With the characteristics of lasting fragrance, it is suitable for cosmetics, detergents and edible essence
=============================================================================
synthetic route
Mainly prepared by O-alkylation reaction of 2-naphthol, common methods include:
Sulfuric acid catalyzed etherification method (classical method)
Dissolve 2-naphthol in anhydrous ethanol, add concentrated sulfuric acid dropwise, and heat to reflux for 10 hours.
The reaction solution was poured into a 5% sodium hydroxide solution to precipitate off white crystals. The crystals were filtered, washed until neutral, and dried to obtain 2-ethoxynaphthalene with a yield of approximately 96%.
Crude products can be refined by vacuum distillation (collecting fractions at 138-140 ° C/1.6 kPa)
Phase transfer catalysis method (green and efficient)
Reaction of 2-naphthol sodium salt with halogenated ethane (such as iodoethane) or diethyl sulfate in the presence of phase transfer catalysts (such as tetrabutylammonium bromide, polyethylene glycol) in a water/organic solvent two-phase system
Mild reaction conditions and high yield (94-98%)
Other methods
Dehydration synthesis of ethanol and 2-naphthol under the action of molecular sieve or solid acid catalyst
Using ionic liquids (such as [BMIM] [PF ?]) as solvents to improve reaction efficiency
