Vanillyl ethyl ether
-
- Category :
Organic chemicals and Derivatives/Aroma compounds
- CAS NO : 13184-86-6
- EC NO : 236-136-4
- Molecular Formula : C10H14O3
- Main Specifications : 99% min
- Synonyms : 236-136-4;alpha-Ethoxy-2-methoxy-P-cresol;Ethyl 4-hydroxy-3-methoxybenzyl ether;Ethyl vanillyl ether;P-Cresol, alpha-ethoxy-2-methoxy-;phenol, 4-(ethoxymethyl)-2-methoxy-;Vanillyl ethyl ether;
Package: 25kg����,50kg 200kg
Uses : Mainly used as a food and daily chemical spice
Molecular Structure:
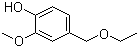
Product description:
What is the chemical of Vanillyl ethyl ether ?
Appearance: Colorless to pale yellow transparent oily liquid ;
Assay:99%min by GC ;
IR Identity: conform to standard ;
HNMR: conform to standard ;
carbon spectrum: conform to standard ;
Water by K. F.:0.5% max or as per the customer’s request ;
Loss on drying:0.5% max. or as per the customer’s request ;
boiling point: 275.2°C / 760 mmHg;
===============================================================================
Purpose:
Mainly used as a food and daily chemical spice, with complex aromas such as creamy vanilla, chocolate, rum, etc
be applied to:
Baked products, cold drinks, gummies, and gummy candies
Dairy products, instant coffee, alcoholic beverages
FEMA usage limit (mg/kg):
Baked products and snacks: 5.0-15
Cold drinks, non-alcoholic beverages, and alcoholic beverages: 3.0~5.0
Soft Candy and Breakfast Grains: 5.0-10
Dairy products: 2.0~5.0
Sugar and icing: 5.0-20
Gum sugar: 10-20
Instant coffee and tea: 3.0 to 10
==============================================================================
Synthetic route
According to the chemical structure (hydroxyl of vanillol is etherified with ethyl), typical synthesis methods may include:
Williamson ether synthesis method: Vanillin alcohol (4-hydroxy-3-methoxybenzyl alcohol) reacts with bromoethane under alkaline conditions.
Alternatively, etherification can be carried out between vanillin and ethanol using dehydration catalysts such as p-toluenesulfonic acid.
