Valerophenone
-
- Category :
Intermediates/Pharmaceutical intermediates
- CAS NO : 1009-14-9
- EC NO : 213-767-3
- Molecular Formula : C11H14O
- Main Specifications : 99% min
- Synonyms : 1-Phenyl-1-pentanone;n-Butyl phenyl ketone;Pentanophenone;
Package: 25kg�����,50kg 200kg
Uses : Phenylacetone is a versatile organic compound, with main applications including: Organic synthesis intermediate: Used for synthesizing complex organic molecules such as drugs, fragrances, and pesticides.
Molecular Structure:
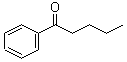
Product description:
What is the chemical of Valerophenone ?
Appearance: Colorless to pale yellow liquid�����;
Assay:99%min by GC�;
IR Identity: conform to standard�;
HNMR: conform to standard����;
carbon spectrum: conform to standard����;
Water by K. F.:0.5% max or as per the customer’s request;
Loss on drying:0.5% max. or as per the customer’s request��;
==========================================================================
Acetophenone is a versatile organic compound with primary applications including: Organic synthesis intermediates for synthesizing complex organic molecules such as pharmaceuticals, fragrances, and pesticides; Pharmaceutical intermediates serving as key building blocks in the pharmaceutical industry; Fragrance components used in formulating food flavors and personal care fragrances (e.g., perfumes, cosmetics); Pesticide synthesis as intermediates for insecticides, herbicides, and fungicides; Materials science, where it can act as a monomer or additive for polymer materials (e.g., plastics, rubbers); And as a preservative component in the storage and transportation of aldehyde chemicals.
===============================================================================
Pentanone can be prepared via two classical organic synthesis routes, both offering high yields and industrial feasibility:
Route 1: Friedel-Crafts Acylation
Raw Materials: Benzene + n-Pentanoyl Chloride
Catalyst: Anhydrous Aluminum Chloride (AlCl?)
Solvent: Dichloromethane (DCM)
Reaction Conditions:
Temperature: Added dropwise at 0–5 °C, then heated to 40 °C for 2 h reaction
Post-treatment: Quenched with hydrochloric acid in an ice bath, liquid-liquid extraction, organic phase concentrated and reduced-pressure distilled
Yield: Approximately 87% (142 g from 120.5 g n-pentanoyl chloride)
Route 2: Grignard Reaction
Raw Materials: Methyl Benzoate + n-Butyl Grignard Reagent (prepared from n-butyl bromide and magnesium)
Solvent: Anhydrous Tetrahydrofuran (THF)
Reaction Conditions:
Grignard reagent addition time: Approximately 30 min
Reaction time: Approximately 3 h
Post-treatment: Acidic hydrolysis
Yield: Approximately 96% (7.8 g from 0.05 mol methyl benzoate)
Among the two methods, the Friedel-Crafts method is more suitable for industrial production due to readily available raw materials and stable operation; the Grignard method is suitable for small-scale high-purity preparation. (For reference only)
