Pyrazineethanethiol
-
- Category :
Organic chemicals and Derivatives/Aroma compounds
- CAS NO : 35250-53-4
- EC NO :
- Molecular Formula : C6H8N2S
- Main Specifications : 99% min
- Synonyms : 2-(2-Mercaptoethyl)pyrazine;2-(pyrazin-2-yl)ethanethiol;2-Pyrazine ethanethiol;β-Mercaptoethyl pyrazine;Pyrazineethanethiol;2-Pyrazinylethanethiol;
Package: 25kg,50kg 200kg
Uses : used as a food flavoring agent, with a strong sulfur and meat flavor
Molecular Structure:
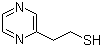
Product description:
What is the chemical of Pyrazineethanethiol ?
Appearance: Colorless to light yellow transparent liquid ;
Assay:99%min by GC ;
IR Identity: conform to standard ;
HNMR: conform to standard ;
carbon spectrum: conform to standard ;
Water by K. F.:0.5% max or as per the customer’s request ;
Loss on drying:0.5% max. or as per the customer’s request ;
boiling point: 105-110 ° C (at 27 hPa or approximately 20 mmHg);
==============================================================================
Usage: 2-Pyrazinylethanethiol is mainly used as a food flavoring agent, with a strong sulfur and meat flavor. It is widely used to simulate and enhance the flavor of the following foods:
beef
chicken
barbecue
coffee
Cooking food with clean water
In addition, it is also used as an organic synthesis intermediate in the production of pharmaceuticals, pesticides, petrochemicals, and dyes. It should be noted that some suppliers clearly label their products as "non medicinal, non edible", limited to industrial or scientific purposes only. In actual application, the product grade (food grade or industrial grade) needs to be selected according to the product level
=============================================================================
Its synthetic route,
1. Reaction of 2-vinylpyrazine with thioacetic acid: This is the mainstream synthetic pathway mentioned by multiple sources. The specific process is as follows: firstly, 2-vinylpyrazine reacts with thioacetic acid (also known as thioglycolic acid) to form an intermediate thiol ester; Subsequently, the thiol ester undergoes conversion steps such as hydrolysis or reduction to ultimately obtain the target product 2-pyrazinoethylthiol.
2. Reaction of 2- (2-chloroethyl) pyrazine with sodium hydrosulfide: Although this route is not directly stated in the provided information, based on its chemical structure (pyrazine ring connected to a - CH ? CH ? SH group) and organic synthesis knowledge, a common synthesis method is through nucleophilic substitution reaction. Using 2- (2-chloroethyl) pyrazine as the starting material, it reacts with sulfur sources such as sodium hydrosulfide (NaSH) to replace the chlorine atom with a thiol group (- SH), thereby generating 2-pyrazinoethylthiol.
In summary, the main synthetic routes of 2-pyrazinoethylthiol (35250-53-4) include:
Route 1: 2-vinylpyrazine+thioacetic acid → intermediate thiol ester → 2-pyrazinoethylthiol
Route 2: 2- (2-chloroethyl) pyrazine+NaSH → 2-pyrazinoethylthiol
