Propyl 4-hydroxybenzoate
-
- Category :
Organic chemicals and Derivatives/Aroma compounds
- CAS NO : 94-13-3
- EC NO : 202-307-7
- Molecular Formula : C10H12O3
- Main Specifications : 99% min
- Synonyms : Propylparaben;Parabens;4-Hydroxybenzoic acid propyl ester;Propyl chemsept;protaben p;pulvis conservans;Solbrol P;tegosept p;4-hydroxybenzoic propyl ester;p-hydroxy propyl benzoate;aseptoform p;betacide p;bonomold op;Chemacide PK;Chemocide PK;Chemoside PK;nipagin p;Nipasol;nipasol m;nipasol p;nipazol;n-Propyl paraben;n-propyl p-hydroxybenzoate;paseptol;preserval p;propagin;Propyl paraben;Propyl Parasept;propyl aseptoform;propyl butex;Propyl Chemosept;Propyl-P-Hydroxybenzoate;Propyl-P-Hydroxy Benzoate;Propyl Nipasol;Propyl Hydroxybenzoate;Propyl p-hydroxybenzoate;
Package: 25kg 50kg drum or bag
Uses : used as a preservative and fungicide in food, cosmetics, pharmaceuticals, and feed, with antibacterial and antioxidant effects.
Molecular Structure:
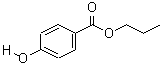
Product description:
What is the chemical of Propyl 4-hydroxybenzoate?
Appearance: White crystalline or crystalline powder, with a special odor, almost odorless, slightly astringent. It is easily soluble in organic solvents such as ethanol, ether, and acetone, and slightly soluble in water ;
Assay: 99% min by HPLC ;
IR Identity: conform to standard ;
HNMR: conform to standard ;
carbon spectrum: conform to standard ;
Water by K. F.:0.5% max or as per the customer’s request ;
Loss on drying:0.5% max. or as per the customer’s request ;
Melting point: 95–98 °C;
==============================================================================
Purpose:
It is mainly used as a preservative and fungicide in food, cosmetics, pharmaceuticals, and feed, with antibacterial and antioxidant effects.
As a bacteriostatic preservative in pharmaceuticals and cosmetics, its preservative effect is greater than that of nipagin ethyl ester.
Used as organic synthesis reagents and pharmaceutical industry raw materials.
It can be mixed with other preservatives such as methylparaben to enhance its antibacterial effect.
==========================================================
Synthesis route: It is prepared by esterification reaction of p-hydroxybenzoic acid and n-propanol under acidic conditions (such as sulfuric acid or cation exchange resin catalysis). After the reaction, the finished product is obtained through steps such as neutralization, washing, decolorization, and recrystallization.
