Perilla aldehyde
-
- Category :
Organic chemicals and Derivatives/Aroma compounds
- CAS NO : 2111-75-3
- EC NO : 243-843-1;218-302-8
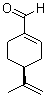
- Molecular Formula : C10H14O
- Main Specifications : 99% min
- Synonyms : L-4(1-Methylethenyl)-1-cyclohexene-1-carboxaldehyde;(-)-Perillaaldehyde;1-methoxy-2,3,5-trimethylbenzene;(4S)-4-(1-methylethenyl)cyclohex-1-ene-1-carbaldehyde;(-)-Perillaldehyde;L-perillaldehyde;;4-isopropenylcyclohex-1-enecarbaldehyde;4-(prop-1-en-2-yl)cyclohex-1-ene-1-carbaldehyde;Perilla aldehyde;
Package: 25kg,50kg 200kg
Uses : as a temporarily permitted flavor
Molecular Structure:
Product description:
What is the chemical of Perilla aldehyde?
Appearance: Colorless to light yellow oily transparent liquid with a strong fat spicy, oily herbal odor ;
Assay:99%min by GC ;
IR Identity: conform to standard ;
HNMR: conform to standard ;
carbon spectrum: conform to standard ;
Water by K. F.:0.5% max or as per the customer’s request ;
Loss on drying:0.5% max. or as per the customer’s request ;;
boiling point: at 104-105 ° C under low pressure conditions of 10 mm Hg;
===============================================================================
Purpose:
Spices: According to the Chinese national standard GB 2760-1996, perillaldehyde is listed as a temporarily permitted flavor, which is mainly used to prepare spices and peanut essence, giving food a unique aroma. FEMA (American Flavor and Extract Manufacturers Association) has clear limits on its use in food, such as 4.5 mg/kg in baked goods and 6.3 mg/kg in frozen dairy products.
Essence&Fragrance Industry: It is widely used to prepare jasmine, narcissus and other floral essence, as well as lemon, spearmint and other fragrance types, and is used in cosmetics and daily chemical products.
Pharmaceutical and chemical intermediates: As a pharmaceutical intermediate, it is used to synthesize other compounds, such as downstream products like L-perilol.
Natural source: It naturally exists in perilla oil, with a content of up to 50%. The crude oil is obtained by steam distillation of perilla whole plant, and then refined and purified by distillation
========================================================================
Main synthetic routes
Extraction and transformation from natural sources:
Perilla frutescens is naturally present in the essential oils of plants such as Perilla frutescens. In industry, a common method is to extract essential oil from perilla leaves through steam distillation, and then enrich and purify perilla aldehyde through fractionation or chromatographic separation techniques. This method is called 'natural extraction', not chemical synthesis, but it is still an important production pathway.
Chemical synthesis route (based on p-menthane skeleton):
The chemical synthesis of perilla aldehyde usually starts from p-Menthane compounds and introduces aldehyde groups through oxidation reactions. A typical route is as follows:
Starting materials: p-Menthene or Limonene, a rich natural monoterpene.
Key step: Selective oxidation of menthene or limonene under specific catalysts (such as transition metal catalysts). This reaction requires precise control of conditions to oxidize specific allyl carbon atoms to aldehyde groups, rather than over oxidation to carboxylic acids.
Product: After oxidation and purification, the racemic form of perilla aldehyde (dl Perillaldehyde, CAS 2111-75-3) is obtained.
The chemical essence of this route is: C10H16 (p-menthone/limonene)+[O] → C10H14O (perilla aldehyde)+H2O.
Chiral synthesis (preparation of a single enantiomer):
The above chemical synthesis usually yields racemic compounds (dl mixtures). To obtain a single chiral isomer, such as (S) - (-) - perilla aldehyde (also known as L-perilla aldehyde), asymmetric synthesis or chiral separation techniques are required. For example, using chiral catalysts for asymmetric hydrogenation or oxidation, or chiral chromatographic separation of dl mixtures.
