Monoethyl Adipate
-
- Category :
Intermediates/Pharmaceutical intermediates
- CAS NO : 626-86-8
- EC NO : 210-966-7
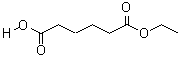
- Molecular Formula : C8H14O4
- Main Specifications : 99% min
- Synonyms : ethyl hydrogen adipate;Monoethyl Adipate;6-ethoxy-6-oxohexanoate;6-ethoxy-6-oxohexanoic acid;MEA;
Package: 25kg 50kg drum or bag
Uses : Organic synthesis intermediates
Molecular Structure:
Product description:
What is the chemical of Monoethyl Adipate ?
Appearance: White or micro powder red crystalline solid ;
Water solubility: Slightly soluble or partially soluble in water (due to the presence of free carboxyl groups)
Easy to dissolve in organic solvents such as alcohols and ethers
Assay: 99% min by HPLC ;
IR Identity: conform to standard ;
HNMR: conform to standard ;
carbon spectrum: conform to standard ;
Water by K. F.:0.5% max or as per the customer’s request ;
Loss on drying:0.5% max. or as per the customer’s request ;
Melting point: 28~29℃;
boiling point: 285 ℃ (atmospheric pressure, 760 mmHg);
====================================================================
Main purpose
Organic synthesis intermediates: used for the preparation of diethyl adipate, 1,6-hexanediol, etc
Pharmaceutical intermediates: such as those used for synthesizing vitamin drugs such as lipoic acid and lipoamide
Solvent: Used for dissolving resins, paints, coatings, and polymers
Plasticizer precursor: modifying material properties in the plastics and resin industry
Chemical reagents: widely used in scientific research and laboratory synthesis
====================================================================
Synthetic route
The standard synthesis method is not detailed in the public information, but according to its structure (HOOC - (CH ?) ? - COOCH ? CH ∝), common synthesis pathways include:
Esterification reaction: Adipic acid and ethanol undergo monoesterification under acid catalysis (such as concentrated sulfuric acid), controlling the reactant ratio (adipic acid: ethanol ≈ 1:1) to preferentially generate monoethyl ester.
Selective hydrolysis/ester exchange: achieved through partial hydrolysis of diethyl adipate or controlled ester exchange with ethanol/water.
Continuous reaction separation process may be adopted in industry to improve monoester selectivity
