Methyl cyclohexanecarboxylate
-
- Category :
Intermediates/Pharmaceutical intermediates
- CAS NO : 4630-82-4
- EC NO : 225-050-2
- Molecular Formula : C8H14O2
- Main Specifications : 99% min
- Synonyms : Cyclohexanecarboxylic acid, methyl ester;4-09-00-00017 (Beilstein Handbook Reference);AI3-04803;BRN 1306359;FEMA No. 3568;Hexahydrobenzoic acid methyl ester;Methyl cyclohexanecarboxylate;Methyl cyclohexanoate;Methyl cyclohexylcarboxylate;Methyl cyclohexylformate;Methyl hexahydrobenzoate
Package: 25kg��,50kg 200kg
Uses : Organic synthesis intermediates
Molecular Structure:
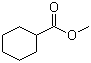
Product description:
What is the chemical of Methyl cyclohexanecarboxylate ?
Appearance: Colorless to pale yellow transparent liquid;
Odor: It has fruity, ester, pineapple and milk like aromas (10% concentration in propylene glycol) ;
Assay:99%min by GC ;
IR Identity: conform to standard ;
HNMR: conform to standard ;
carbon spectrum: conform to standard ;
Water by K. F.:0.5% max or as per the customer’s request ;
Loss on drying:0.5% max. or as per the customer’s request ;
boiling point: 183 ° C (atmospheric pressure, 760 mmHg);
==============================================================================
Purpose:
Organic synthesis intermediates: used for synthesizing drugs, fragrances, and other fine chemicals
Food spices: comply with GB 2760-1997 regulations, allowed to be used as edible spices to impart flavors to fruits, cream, etc
Essence formula: widely used in fruity and frankincense essence, such as pineapple, milk, apple and other flavors
=============================================================================
synthetic route
Methyl cyclohexane can be prepared by the following main methods:
Fischer esterification method with cyclohexane carboxylic acid and methanol acid catalysis (such as H2SO4), reflux ≈ 99%
The reaction between methyl formate and cyclohexene addition in the presence of cyclohexene+methyl formate catalyst is approximately 48%
3-Chlorocyclohexane two-step method: Chlorocyclohexane → hydrolysis → esterification hydrolysis to obtain cyclohexanol, followed by oxidation esterification with a total yield of 65%
Note: Fischer esterification method is a mainstream process in laboratory and industry, with simple operation and high yield.
