M-Anisaldehyde
-
- Category :
Organic chemicals and Derivatives/Aroma compounds
- CAS NO : 591-31-1
- EC NO : 209-712-8
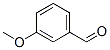
- Molecular Formula : C8H8O2
- Main Specifications : 99% min
- Synonyms : 3-methoxybenzaldehde;3-methoxy-benzaldehyd;Metamethoxybenzaldehyde;M-METHOXYBENZALDEHYDE;AKOS BBS-00003247;3-ANISALDEHYDE;META-ANISALDEHYDE;ANISALDEHYDE,3-;META-ANISALDEHYD;m-Methoxybenzaldehyde;M-Anisaldehyde;3-Anisaldehyd;Benzaldehyde, 3-methoxy-;3-methoxybenxaldehyde;M-ANISADEHYDE;phenanthro[1,2-b]thiophene;3-Methoxybenzyaldehyde;
Package: 25kg��,50kg 200kg
Uses : M-Anisaldehyde is widely used as an intermediate in the synthesis of flavorings, pharmaceuticals, pesticides, dyes, and daily chemical products.
Molecular Structure:
Product description:
What is the chemical of M-Anisaldehyde ?
Appearance: Colorless or pale yellow oily liquid�;
Assay:99%min by GC��;
IR Identity: conform to standard�;
HNMR: conform to standard�;
carbon spectrum: conform to standard;
Water by K. F.:0.5% max or as per the customer’s request����;
Loss on drying:0.5% max. or as per the customer’s request����;
===============================================================================
M-Anisaldehyde is widely used as an intermediate in the synthesis of flavors, pharmaceuticals, pesticides, dyes, and daily chemical products. In flavors, it serves as a main or modifier component in floral types (such as hawthorn, lilac, sunflower, osmanthus) and citrus essences. As an organic intermediate, it is extensively applied in the synthesis of pharmaceuticals, pesticides, dyes, and daily chemicals. For pharmaceutical raw materials, it acts as a synthetic precursor for certain drugs (e.g., veterinary medicines and livestock/poultry medications). In research, it is commonly used as an aldehyde substrate in organic synthesis reactions.
收率較高,適合實驗室制備 ?(僅供參考)
==============================================================================
The main synthetic methods include:
Methylation (reaction of m-hydroxybenzal-dehyde with dimethyl sulfate)
Reaction conditions: alkaline environment, mild heating
Advantages: readily available raw materials, simple operation
Oxidation method (using m-methoxybenzyl alcohol as the raw material)
Oxidizing agent: nitric acid (molar ratio 1.5:1)
Process: nitric acid is added dropwise at low temperature (<5°C), reaction proceeds for about 7 hours; post-treatment includes neutralization, extraction, drying, and vacuum distillation
High yield, suitable for laboratory preparation (for reference only)
