Ionone apha
-
- Category :
Organic chemicals and Derivatives/Aroma compounds
- CAS NO : 127-41-3
- EC NO :
- Molecular Formula : C13H20O
- Main Specifications : 99% min
- Synonyms : ionone;(3E)-4-[(1S)-2,6,6-trimethylcyclohex-2-en-1-yl]but-3-en-2-one;(3E)-4-(2,6,6-trimethylcyclohex-2-en-1-yl)but-3-en-2-one;(3E)-4-[(1R)-2,6,6-trimethylcyclohex-2-en-1-yl]but-3-en-2-one;4-(2,6,6-trimethylcyclohex-1-ene-1-yl)-but-3-ene-2-one;4-(2,6,6-trimethylcyclohex-1-en-1-yl)but-3-en-2-one;(3E)-4-(2,6,6-trimethylcyclohex-1-en-1-yl)but-3-en-2-one;(3Z)-4-(2,6,6-trimethylcyclohex-1-en-1-yl)but-3-en-2-one;α-Ionone;Irisone;Ionone apha;
Package: 25kg��,50kg 200kg
Uses : in daily chemical essence, edible spices
Molecular Structure:
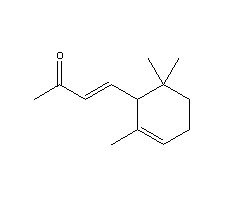
Product description:
What is the chemical of Ionone apha?
Appearance: Colorless to slightly yellow transparent liquid
Odor: Strong violet aroma, accompanied by warm woody notes, iris roots, and fruity notes; Diluted to produce iris root fragrance, mixed with ethanol to enhance violet fragrance;
Assay:99%min by GC ;
IR Identity: conform to standard ;
HNMR: conform to standard ;
carbon spectrum: conform to standard ;
Water by K. F.:0.5% max or as per the customer’s request ;
Loss on drying:0.5% max. or as per the customer’s request ;
boiling point: 259–263 °C(lit.);
===============================================================================
Main purpose
Daily chemical essence: used for the preparation of essence such as soap and cosmetics
Edible spices: According to the Chinese national standard GB 2760-96, they are temporarily allowed to be used as edible spices for the preparation of aroma types such as longan, raspberry, blackberry, cherry, citrus, etc
Other uses: Biochemical research, organic synthesis raw materials
==============================================================================
Synthetic route
In industry, it is mainly synthesized through the following two-step method
Condensation reaction: Citral (derived from Shan Cangzi oil, containing about 70% citral) is condensed with acetone in the presence of sodium hydroxide to produce pseudo violet ketone.
Cyclization reaction: Pseudo violet ketone is heated and cyclized in dilute sulfuric acid/glycerol solution to obtain a mixture of α - violet ketone (10-20%) and β - violet ketone (80-90%), which is then separated by distillation to obtain the target product.
