Hexyl valerate
-
- Category :
Organic chemicals and Derivatives/Aroma compounds
- CAS NO : 1117-59-5
- EC NO : 214-248-4
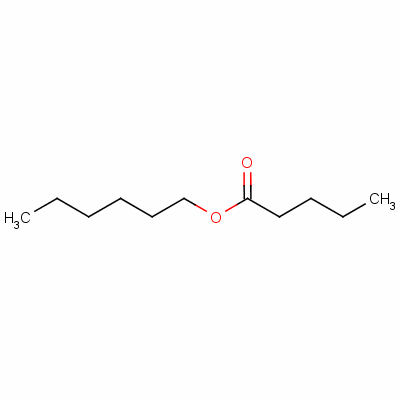
- Molecular Formula : C11H22O2
- Main Specifications : 99% min
- Synonyms : Pentanoic acid, hexyl ester;1-Hexyl n-valerate;AI3-30516;Hexyl pentanoate;Hexyl valerate;Hexyl valerianate;NSC 21861;Valeric acid, hexyl ester (8CI)
Package: 25kg,50kg 200kg
Uses : Used extensively as an intermediate in flavorings, fragrances, pharmaceuticals, and organic solvents.
Molecular Structure:
Product description:
What is the chemical of Hexyl valerate?
Appearance: Colorless transparent liquid���;
Assay:99%min by GC;
IR Identity: conform to standard;
HNMR: conform to standard��;
carbon spectrum: conform to standard���;
Water by K. F.:0.5% max or as per the customer’s request���;
Loss on drying:0.5% max. or as per the customer’s request�����;
=============================================================================
Uses: As an intermediate, it is widely used in flavor and fragrance, pharmaceuticals, organic solvents, etc.
Flavor and Fragrance: Widely used in food and daily chemical products to provide fruity notes (such as apple, banana, etc.);
Drug Analysis: Used as a derivatization reagent or internal standard for the development of detection methods for drug metabolites;
Organic Synthesis Intermediate: Used for structural modification and reaction research of ester compounds;
Industrial Solvent: Used as a solvent of moderate polarity in certain specific systems.
===============================================================================
n-Hexyl valerate is typically prepared via esterification, mainly through the following two methods:
Fischer Esterification (Acid-Catalyzed Esterification):
- Reactants: Valeric acid + 1-Hexanol
- Catalyst: Concentrated sulfuric acid or p-toluenesulfonic acid
- Conditions: Heated under reflux with removal of generated water to drive the equilibrium
Acyl Chloride-Alcohol Method (Suitable for High Purity Requirements):
- Steps: First convert valeric acid into n-valeryl chloride, then react with 1-hexanol under basic conditions
- Advantages: Fast reaction rate, high yield, and fewer by-products
The first method is predominantly used in industry due to its readily available raw materials and lower cost. (For reference only)
