Furfuryl mercaptan
-
- Category :
Organic chemicals and Derivatives/Aroma compounds
- CAS NO : 98-02-2
- EC NO : 202-628-2
- Molecular Formula : C5H6OS
- Main Specifications : 99% min
- Synonyms : 2-Furanmethanethiol;2-furylmethanethiol;furan-2-ylmethanethiol;
Package: 25kg,50kg 200kg
Uses : As a food spice, it is widely used to make essence
Molecular Structure:
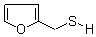
Product description:
What is the chemical of Furfuryl mercaptan ?
Appearance: Colorless to pale yellow transparent oily liquid with a strong and unpleasant sulfur odor; But at extremely low concentrations, it can present strong coffee and meat aromasd ;
Assay:99%min by GC ;
IR Identity: conform to standard ;
HNMR: conform to standard ;
carbon spectrum: conform to standard ;
Water by K. F.:0.5% max or as per the customer’s request ;
Loss on drying:0.5% max. or as per the customer’s request ;
Boiling point: 155 ° C (at 760 mmHg atmospheric pressure);
==============================================================================
Purpose:
As a food spice, it is widely used to make essence of coffee, beef, onion, garlic, chocolate, tobacco, etc.
According to the Chinese standard GB 2760-1996, it is allowed to be used as a food additive, and the maximum allowable dosage varies depending on the food category (such as 0.52 mg/kg in soft drinks and 2.0 mg/kg in candies).
Used as an intermediate in organic synthesis for the preparation of thioether compounds, pesticides (such as methyl phosphate), etc.
=============================================================================
synthetic route
The main industrial synthesis routes for furfuryl mercaptan are as follows:
Hydrolysis after reaction of furfuryl chloride with thiourea:
Furfuryl chloride (2-chloromethylfuran) reacts with thiourea to form thiourea adducts, which are then subjected to alkaline hydrolysis to obtain furfuryl mercaptan.
Reaction of furfuryl alcohol with thiourea and hydrobromic acid:
Furfuryl alcohol is heated and refluxed with thiourea in the presence of hydrobromic acid to form an intermediate, which is then treated with sodium hydroxide, acidified, extracted, and purified by distillation to obtain the product.
It can be prepared by multi-step reactions such as reduction, halogenation, and thiolation of 2-furanaldehyde (furfural), but the first route is mainly used in industry.
