Ethyl thioacetate
-
- Category :
Organic chemicals and Derivatives/Aroma compounds
- CAS NO : 625-60-5
- EC NO : 261-601-3;210-904-9
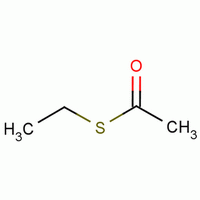
- Molecular Formula : C4H8OS
- Main Specifications : 99% min
- Synonyms : Ethanethioic acid, ethyl ester;Acetic acid, thio-, ethyl ester;Ethyl ethanethioate;S-ethyl ethanethioate;S-Ethyl thioacetate;Thioacetic acid S-ethyl ester;O-ethyl ethanethioate;S-ethyl ethanethoate;S-ethylthioacetate;Ethanethioic Acid S-Ethyl Ester;
Package: 25kg,50kg 200kg
Uses : used as a food flavor
Molecular Structure:
Product description:
What is the chemical of Ethyl thioacetate?
Appearance: Colorless to pale yellow clear liquid, the odor has a strong foul odor;
Assay:99%min by GC ;
IR Identity: conform to standard ;
HNMR: conform to standard ;
carbon spectrum: conform to standard ;
Water by K. F.:0.5% max or as per the customer’s request ;
Loss on drying:0.5% max. or as per the customer’s request ;
boiling point: 116 °C;
===========================================================================
Use: It is mainly used as a food flavor (flavor enhancer) to provide sulfur, fruit flavor, onion, garlic, meat quality, coffee and other flavor characteristics in the essence formula
Food industry: used as an edible spice to enhance the flavor characteristics of beer, white wine, red wine, and rose wine.
Essence formula: widely used in fragrance mixing system, giving complex fragrance such as spice, meat and milk, meeting FEMA 3282 standard.
Chemical synthesis: As an intermediate of thioesters, it is used to introduce ethylthio functional groups in organic synthesis
==========================================================================
synthetic route
Standard laboratory synthesis method (based on literature):
reactant:
Acetyl chloride (1.0 equivalent)
Thiol (1.2 equivalents)
Triethylamine (2.0 equivalents)
4-Dimethylaminopyridine (DMAP, 0.05 equivalents)
Solvent: Dichloromethane (DCM)
Reaction conditions:
Temperature: Slowly drip acetyl chloride at 0 ° C
Heat up to room temperature and continue stirring
TLC monitoring of reaction progress (approximately 2-4 hours)
post-processing
Filter to remove salt precipitates
Reduced pressure concentrated filtrate
Rapid column chromatography purification (silica gel, petroleum ether/ethyl acetate elution)
Yield: The literature is not clear, but it belongs to a highly selective reaction, and the yield is usually>80%
Attention: This reaction should be carried out in a fume hood to avoid contact with thiol odor substances and strong irritant vapors of acetyl chloride.
