Citronellal
-
- Category :
Organic chemicals and Derivatives/Aroma compounds
- CAS NO : 106-23-0
- EC NO : 203-376-6
- Molecular Formula : C10H18O
- Main Specifications : 99% min
- Synonyms : 3,7-Dimethyl-6-octenal;Natural Citronellal;3,7-dimethyloct-6-enal;(4-chlorophenyl)(phenyl)methanone;(3R)-3,7-dimethyloct-6-enal;
Package: 25kg�����,50kg 200kg
Uses : Synthetic raw materials
Molecular Structure:
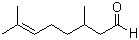
Product description:
What is the chemical of Citronellal?
Appearance: Colorless to slightly yellow liquid with strong lemon, lemongrass, and rose like aromas. It exists in three forms of optical rotation: d -, l -, and dl -, among which d-citral is abundant in natural essential oils. ;
Assay:99%min by GC ;
IR Identity: conform to standard ;
HNMR: conform to standard ;
carbon spectrum: conform to standard ;
Water by K. F.:0.5% max or as per the customer’s request ;
Loss on drying:0.5% max. or as per the customer’s request ;
boiling point: 203-208 ° C, measured at 760 mmHg;
==============================================================================
Purpose:
Essence and spice: GB 2760-96 stipulates that it is a permitted food spice, which is mainly used to prepare citrus and cherry essence, as well as low-grade lemon, cologne, soap essence, etc., to provide outstanding grass flavor.
three
Synthetic raw materials: are key intermediates for the synthesis of important fragrances such as citronellol, hydroxycitronellal, and menthol.
Other applications: Used for pharmaceutical synthesis, daily chemical products (such as skincare and cosmetics), and seasonings.
===============================================================================
Synthesis route:
Natural extraction: Obtained from natural essential oils such as lemongrass oil and lemon eucalyptus oil through methods such as distillation and purification with sodium bisulfite addition. Chemical synthesis:
Catalytic hydrogenation of citral: The main method in industry is to produce citral by hydrogenation of citral under the action of a catalyst.
Catalytic dehydrogenation of citronellol: In the presence of lead chromium catalyst, citronellol can be dehydrogenated to form citral.
Other methods: such as preparing from pinene through multi-step reactions, or hydrogenating citral in isopropanol under the action of nickel formate/alumina catalyst.
