Benzanthrone
-
- Category :
Dyestuffs and Pigments
- CAS NO : 82-05-3
- EC NO : 201-393-3
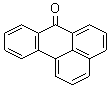
- Molecular Formula : C17H10O
- Main Specifications : 99% min
- Synonyms : 7H-Benz[de]anthracen-7-one;Benzanthrone,98%;benz(d e)anthracen-7-one;7H-benzo[de]anthracen-7-one;
Package: 25kg 50kg drum or bag
Uses : The main use of Benzanthrone is as a dye intermediate for producing various vat dyes, such as: Vat Brilliant Green FFB; Vat Olive Green B; Vat Olive T; Vat Gray M; Vat Black BBN. Benzanthrone is also used as a sensitizer in degradable thermoplastic produc
Molecular Structure:
Product description:
What is the chemical of Benzanthrone ?
Appearance: Light yellow needle-like crystals or powder;
Assay:99%min by HPLC��;
IR Identity: conform to standard�;
HNMR: conform to standard����;
carbon spectrum: conform to standard�����;
Water by K. F.:0.5% max or as per the customer’s request;
Loss on drying:0.5% max. or as per the customer’s request����;
===============================================================================The main use of Benzanthrone is as a dye intermediate for producing various vat dyes, such as: Vat Brilliant Green FFB; Vat Olive Green B; Vat Olive T; Vat Gray M; Vat Black BBN. Benzanthrone is also used as a sensitizer in degradable thermoplastic products.
===============================================================================
Synthesis Route: The synthesis of benzanthrone typically starts with anthraquinone and glycerol, undergoing the following steps: Reduction: Anthraquinone is reduced with iron powder in the presence of copper sulfate to form hydroxyanthraquinone; Dehydration: Glycerol undergoes dehydration under concentrated sulfuric acid to produce acrolein; Condensation: Hydroxyanthraquinone reacts with acrolein in the presence of sulfuric acid to undergo a condensation reaction, losing one molecule of water to form a new ring; Oxidation: Finally, the intermediate is oxidized by sulfuric acid to yield benzanthrone. Other synthesis methods include C-H bond activation and cyclization reactions using palladium catalysts, or oxidation of benzophenanthrene derivatives, among other pathways.
