Acetanisole
-
- Category :
Organic chemicals and Derivatives/Aroma compounds
- CAS NO : 70-70-2
- EC NO : 200-743-2
- Molecular Formula : C9H10O2
- Main Specifications : 99% min
- Synonyms : Paroxypropione;p-Methylbenzoic Acid Methyl Ether;P-Hydroxypropiophenone;P-Hydroxyl propiophenone;P-Methoxyacetophenone;PARA Methoxy acetophenone;VANATONE;1-(4-hydroxyphenyl)-1-propanon;1-(4-Hydroxyphenyl)-1-propanone;(4-Hydroxyphenyl)-1-propanone;1-(4-hydroxyphenyl)propan-1-one;4-Hydroxypropiophenone;4'-Hydroxy propiophenone;
Package: 25kg 50kg drum or bag
Uses : Mainly used as an intermediate in organic synthesis
Molecular Structure:
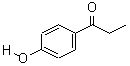
Product description:
What is the chemical of Acetanisole ?
Appearance: White needle shaped or prismatic crystals ;
Assay: 99% min by HPLC ;
IR Identity: conform to standard ;
HNMR: conform to standard ;
carbon spectrum: conform to standard ;
Water by K. F.:0.5% max or as per the customer’s request ;
Loss on drying:0.5% max. or as per the customer’s request ;
Melting point: 36–38 °C;
==============================================================================
Usage: Mainly used as an intermediate in organic synthesis, especially in the preparation of liquid crystal materials, it has important applications. In addition, it is also used for the synthesis of pharmaceutical intermediates and other fine chemicals
==============================================================================
synthetic route
There are multiple synthetic routes for 4 '- hydroxyphenylacetone, and the following are some publicly available routes:
Using 4-hydroxyacetophenone (121-97-1) as the raw material, it was synthesized through alkylation or reductive carbonylation reactions, with a reported yield of approximately 68% in the literature.
Starting from 4-hydroxyBenzoic aldehyde (637-27-4), acetone groups were constructed through Clemson reduction or Wittig reaction with a yield of approximately 58%.
Other routes include using 4-hydroxybenzoic acid derivatives or constructing aromatic ketone structures through Friedel Crafts acylation reactions.
