Acetanilide
-
- Category :
Organic chemicals and Derivatives/Aroma compounds
- CAS NO : 103-84-4
- EC NO : 203-150-7
- Molecular Formula : C8H9NO
- Main Specifications : 99% min
- Synonyms : Acetanilide;Antifebrin;Acetanilide,(N-Phenylacetamide);N-Phenylacetamide;ACTN;
Package: 25kg 50kg drum or bag
Uses : an important organic synthesis intermediate
Molecular Structure:
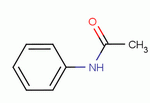
Product description:
What is the chemical of Acetanilide?
Appearance: White glossy flaky crystals or white crystalline powder, odorless, but may have a slight burnt smell. Stable in air, but sensitive to light ;
Assay: 99% min by HPLC ;
IR Identity: conform to standard ;
HNMR: conform to standard ;
carbon spectrum: conform to standard ;
Water by K. F.:0.5% max or as per the customer’s request ;
Loss on drying:0.5% max. or as per the customer’s request ;
Melting point: 113–115 °C;
boiling point: 304 ° C (at standard atmospheric pressure);
===============================================================================
purpose
N-acetyl-ani-line is an important organic synthesis intermediate, mainly used for:
Pharmaceutical industry: As a raw material for sulfonamide drugs such as sulfamethoxazole, it has been historically used as an analgesic and antipyretic (but has been replaced by acetaminophen due to its toxicity).
Dye industry: used for manufacturing dye intermediates such as p-nitroacetanilide, p-nitroani-line, and p-phenylenediamine.
Industrial applications: Used as a rubber vulcanization accelerator, fiber lipid coating, and stabilizer for hydrogen peroxide, as well as for the synthesis of camphor and thioacetamide.
Analytical Chemistry: As an analytical reagent, it is used to detect cerium, chromium, iron, cyanide, lead, manganese, oxidants, nitrates, and nitrites
==============================================================================
synthetic route
N-acetyl-ani-line is mainly prepared through the acetylation reaction of ani-line, and there are two commonly used methods:
Acetic acid acetylation method: Mix ani-line with excess glacial acetic acid, heat and reflux for 6-14 hours, cool and crystallize after the reaction is complete, filter, wash with water, and dry to obtain the product.
Acetic anhydride acylation method: In a benzene solution, ani-line is reacted with excess acetic anhydride under milder reaction conditions, resulting in higher product purity.
