4'-Methoxyacetophenone
-
- Category :
Intermediates/Pharmaceutical intermediates
- CAS NO : 100-06-1
- EC NO : 202-815-9
- Molecular Formula : C9H10O2
- Main Specifications : 99% min
- Synonyms : p-Acetanisole;p-Acetylanisole;4-Methoxyacetophenone;4-Methoxy Acetophenone;1-(4-methoxyphenyl)ethanone;P-methoxy acetophone;Acetanisole;
Package: 25kg 50kg drum or bag
Uses : Spice industry
Molecular Structure:
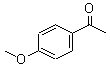
Product description:
What is the chemical of 4'-Methoxyacetophenone?
Appearance: White to slightly yellow crystalline or crystalline powder;
Odor: It has a sweet anise bean aroma, with floral notes such as hawthorn flowers and mimosa flowers
Solubility: Slightly soluble in water (solubility at 20 ° C is 2.474 g/L), easily soluble in organic solvents such as ethanol, ether, acetone, etc
Sensitivity: Sensitive to light and should be stored away from light Assay: 99% min by HPLC ;
IR Identity: conform to standard ;
HNMR: conform to standard ;
carbon spectrum: conform to standard ;
Water by K. F.:0.5% max or as per the customer’s request ;
Loss on drying:0.5% max. or as per the customer’s request ;
Melting point: 36–38 °C(lit.) ;
===============================================================================
main purpose
Spice industry:
It is used to prepare hawthorn flower, mimosa, acacia, new cut grass and other fragrant essence
Suitable for soap, cosmetics, tobacco, food (such as vanilla beans, cream, nuts, etc.) essence
GB 2760 specifies that edible spices are allowed to be used, FEMA number 2005 56
Organic synthesis:
As an intermediate used in the synthesis of p-methoxyphenylacetic acid (puerarin intermediate), liquid crystal monomers, sunscreen agents, etc
==============================================================================
synthetic route
Methoxyacetophenone can be synthesized by the following methods:
1. Friedel Crafts acetylation reaction (commonly used method):
Raw materials: benzyl ether (anise ether)+acetyl chloride (or acetic anhydride)
Catalyst: Anhydrous aluminum trichloride (AlCl ∝), ferric trichloride (FeCl ∝), or acidic clay
Condition: Under carbon disulfide (CS ?) or direct heating reflux (about 140 ° C), the product is purified by washing with water and vacuum distillation after several hours of reaction
2. Modern green catalytic method (literature report):
Using MoSi composite metal oxide catalyst to catalyze the reaction of benzyl ether and acetyl chloride at 80 ° C for 10 hours, the yield is relatively high
3. Other methods:
Reaction of Benzyl Ether and Acetic Acid in the Presence of Boron Trifluoride (BFVNet)
