4-Heptanone
-
- Category :
Organic chemicals and Derivatives/Aroma compounds
- CAS NO : 123-19-3
- EC NO : 204-608-9
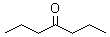
- Molecular Formula : C7H14O
- Main Specifications : 99% min
- Synonyms : Di-n-propyl ketone;DIPROPYL KETONE;HEPTANONE, 2-;FEMA 2546;FEMA 2544;BUTYRONE;METHYL PENTYL KETONE;METHYL N-AMYL KETONE;heptan-4-one;
Package: 25kg,50kg 200kg
Uses : As an edible spice and as an industrial solvent and organic synthesis intermediate
Molecular Structure:
Product description:
What is the chemical of 4-Heptanone ?
Appearance: colorless transparent liquid with low volatility and a fragrant aroma. Its specific smell is described as strong ether and fruit aroma, accompanied by cheese, sweet, cognac, jackfruit and other aroma;
Assay:99%min by GC
IR Identity: conform to standard ;
HNMR: conform to standard ;
carbon spectrum: conform to standard ;
Water by K. F.:0.5% max or as per the customer’s request ;
Loss on drying:0.5% max. or as per the customer’s request ;
boiling point: 144.0±0.0 °C at 760 mmHg;
==============================================================================
Usage: 4-Heptone has two main uses:
As an edible spice: used to prepare jackfruit, cognac, banana, tropical fruit and other types of essence, which naturally exist in apple juice, papaya, pears, baked potatoes, bread, coffee, peanuts and other substances.
As an industrial solvent and organic synthesis intermediate: used as a solvent for nitrocellulose and involved in organic synthesis reactions
===============================================================================
There are several main synthetic routes:
Preparation by decarboxylation of butyric acid: butyric acid is decarboxylated at high temperature (about 425 ℃) using charcoal catalyst, and then at higher temperature (about 500 ℃) using cerium oxide or thorium oxide catalyst to produce 4-heptone.
Through oxidation reaction: Butyric acid can also undergo oxidative dehydrogenation reaction at 400-425 ℃ using an oxidation catalyst (such as oxidative thistle) to directly generate 4-heptanone.
Aldehyde condensation: In industry, n-butyraldehyde or acetaldehyde can also be used for aldol condensation reaction under alkaline conditions, followed by dehydration, hydrogenation and other steps to synthesize 4-heptone.
In addition, 4-heptanone is naturally present in foods such as apple juice, papaya, pears, baked potatoes, bread, and coffee, but commercial use typically relies on the chemical synthesis methods mentioned above
