2-Phenylbutyric acid
-
- Category :
Intermediates/Pharmaceutical intermediates
- CAS NO : 90-27-7
- EC NO : 201-982-5
- Molecular Formula : C26H43NO6
- Main Specifications : 99% min
- Synonyms : 2-Phenylbutyric acidAV23224;2-Phenyl-butyric acid;2-phenyl-butyricaci;alpha-Phenylbutyric acid;alpha-phenylbutyricacid;alpha-Toluic acid, alpha-ethyl-;Butyric acid, 2-phenyl-;RARECHEM AL BO 0093;A-ETHYLPHENYLACETIC ACID;AKOS BBS-00006483;ALPHA-ETHYLPHENYLACETIC ACID;ALPHA-PHENYL-N-BUTYRIC ACID;2-PHENYLBUTANOIC ACID;2-PHENYL-N-BUTYRIC ACID;2-ETHYL-2-PHENYLACETIC ACID;AURORA KA-7220;(2R)-2-phenylbutanoate;(2S)-2-phenylbutanoate;oxo{[(3α,7β,8ξ,9ξ,12α,14ξ)-3,7,12-trihydroxycholan-3-yl]amino}acetic acid;
Package: 25kg 50kg drum or bag
Uses : Mainly used as pharmaceutical intermediates
Molecular Structure:
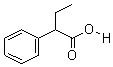
Product description:
What is the chemical of 2-Phenylbutyric acid ?
Appearance: White adhesive crystalline powder with aromatic odor ;
Assay: 99% min by HPLC ;
IR Identity: conform to standard ;
HNMR: conform to standard ;
carbon spectrum: conform to standard ;
Water by K. F.:0.5% max or as per the customer’s request ;
Loss on drying:0.5% max. or as per the customer’s request ;
Melting point: 39–42 °C(lit.);
boiling point: 270–272 °C(lit.);
=============================================================================
Purpose:
Mainly used as pharmaceutical intermediates, such as impurity reference standards for the synthesis of drugs such as amiodarone and pheno-barbital.
As an aromatic substituted fatty acid block in organic synthesis.
Has biological activity against hypercholesterolemia
==============================================================================
Synthesis route (partially disclosed methods)
According to existing literature, the synthesis route of 2-phenylbutyric acid includes:
Reaction of phenylacetic acid with bromoethane:
Phenylacetic acid (103-82-2) undergoes alkylation reaction with bromoethane (74-96-4) under alkaline conditions, yielding approximately 90% yield of 2-phenylbutyric acid.
2-phenylsuccinic anhydride hydrolysis:
Using 2-phenylsuccinic anhydride (1519-21-7) as a precursor, hydrolysis under specific conditions can yield the target product in approximately 96% yield.
Other possible paths:
Including modern organic synthesis methods such as addition hydrolysis of Grignard reagents with nitrile or esters, or transition metal catalyzed C-C coupling (although not detailed in the provided information, it is a common strategy for synthesizing arylalkyl carboxylic acids)
