catalase
-
- Category :
Food and Feed additives/Food additives
- CAS NO : 7722-84-1
- EC NO : 231-765-0
- Molecular Formula : H2O2
- Main Specifications : high quality and technical support, food and industry grade, remove hydrogen peroxide, liquid and powder base, higher activity.
- Synonyms : Hydrogen Peroxide 30% solution;hydrogen peroxide 35%;Hydrogen peroxide solution;Hydrogen peroxide 100 volumes;Industrial Hydrogenperoxide;
Package: 20 kg/Barrel??? 1kg*20bags/box.
Uses : high quality and technical support, food and industry grade, remove hydrogen peroxide, liquid and powder base, higher activity.
Molecular Structure:
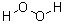
Product description:
General introduction Catalase is a common enzyme present in most all living organisms and a very potent catalyst. Catalase converts the harmful byproduct of metabolism, Hydrogen Peroxide, into water and oxygen. It also oxidizes toxins in the body such as formaldehyde, alcohols, phenols and formic acid. 2H2O2 → O2 + 2H2O Catalase is present in every known animal and is vital to life processes. In addition to being a catalyst, it is also used in various other ways from removing hydrogen peroxide from milk prior to making it into cheese, to contact lens disinfectant and preventing food from oxidizing through its presence in food wrappers. Catalase is an enzyme found in the cells of animals, plants and aerobic bacteria. An enzyme is a large organic molecule synthesized in cells and made to act as a catalyst in a reaction. Each type of enzyme performs a specific function, and the function of catalase is to convert a potentially harmful by-product into usable elements. H2O2 + H2R →2H2O + R Function
Catalase, like most enzymes, is a protein. It is found in perixisomes, which are membrane-bound organelles of cells. Catalase has an important biological function: it catalyzes the breakdown of hydrogen peroxide, a toxic substance to organisms, to water and molecular oxygen, which are both harmless and useful. Structure o Catalase is a dumbbell shaped structure with four polypeptide chains, each containing over 500 amino acids. Catalase also has four heme groups, which are made up of protoporphyrin rings containing a single iron atom. These rings are nestled within the four chains.
The Importance of Catalase Hydrogen peroxide is a by-product of many biological functions of organisms. All aerobic organisms use oxygen for respiration. The reduction of oxygen into water is sometimes incomplete, and an extra electron from a metal ion can be transferred, causing the formation of peroxide. However, most of the hydrogen peroxide is created during the production of ATP in the mitochondria.
Although peroxide is toxic to living cells, the reaction to break it back down to water and oxygen happens quickly and efficiently. Catalase is known as one of the most efficient enzymes, having a turnover number close to 200,000 events/second/subunit. Without catalase to increase the rate of the reactions, hydrogen peroxide would remain, damaging cells.
